If the anion is a simple ion it takes its name from the root name of the element, and -ide is added. However, since we only talk about one polyatomic type II cation this should not prove to be difficult. If the cation is a polyatomic you use the name that you have memorized. Silver cation SILVER ION Silver, ion (Ag1+) silver(1+) 1 Molecular Weight. If the cation is a "simple" cation the cation is simply the name of the element. need to know the name, formula, and charge of each ion as the names and formulas will be. So, the compound is silver(I) chloride, but it is commonly known as silver. Common Ionic Species in Aqueous Solution. For example ferric is Fe 3+ and ferrous is Fe 2+. Refer to the periodic table above, Ag forms 1+ charge and Cl forms 1- charge. A negatively charged ion, which has more electrons in its electron shells than it has protons in its nuclei, is known as an anion (pronounced /nan/ an-eye. ic is for the higher charged cation, -ous is for the lower charged cation. To find the correct oxidation state of Ag in AgNO3 (Silver nitrate), and each element in the molecule, we use a few rules and some simple math.First, since t. Occasionally suffixes, -ic and -ous, are used however, we will not be using them. Transition metals (in the central block between groups 2. Elements in the same group form ions with the same charge. The cation is named first and the charge of the cation is expressed as a Roman numeral. The charge of many ions can be worked out using patterns in the periodic table. Briefly, the first cycle charge/discharge curves confirms the formation of SEI layers at a voltage plateau of approximately 0. If the cation is not from group 1 or 2, or is not Ag +,Cd 2+, Zn 2+, or Al 3+ then assume the cation is a type II cation. The first cycle galvanostatic charge/discharge voltage profiles for PG, AG, and KG at a current density of 50 mA g 1 are also shown in Fig. A chemical formula is a concise list of the elements in a compound and the ratios of these elements. We have already encountered some chemical formulas for simple ionic compounds. Recognize polyatomic ions in chemical formulas. Naming compound with type II cations-type II cations are capable of having several charges. Learning Objectives Write the chemical formula for a simple ionic compound. NaCl, MgCl 2, AlCl 3, NaNO 3, BaSO 4, (NH 4) 2SO 3ī. But you need to know the specific ionic charge elments. Non-metals (found on the right) will be negative. On the Periodic Table metals (found on the left of the table) will be positive. To find the ionic charge of an element youll need to consult your Periodic Table. If the anion is a polyatomic anion you use the name that you have memorized. Finding Charges of Ions on Periodic Table. If the anion is a simple ion it takes its name from the root name of the element, and "-ide " is added. X-ray photoelectron spectroscopy confirmed the co-existence of both metallic silver (Ag 0) and silver ion (Ag +) on the Ag-doped TiO 2 chitosan composite surface due to the oxidation of a little amount of silver nanoparticles on the composite surface as a consequence of the storage process. 
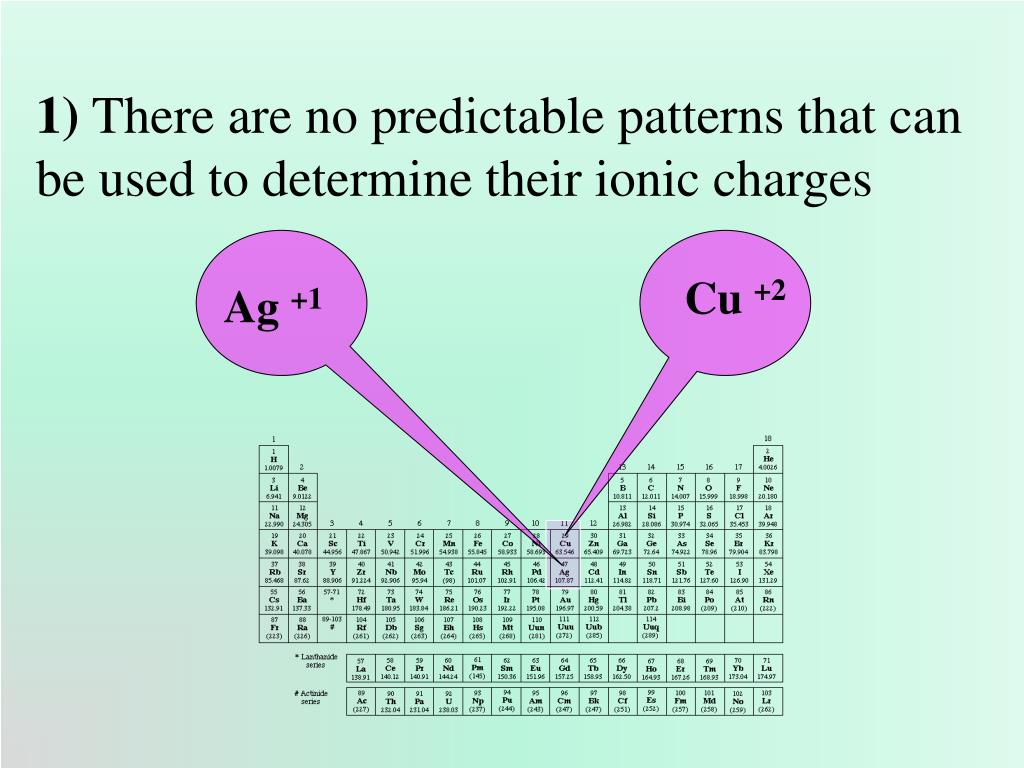
The Hg 2 2+ cation is a polyatomic ion and a type II cation.). If the cation is a polyatomic cation (a polyatomic ion is made from two or more covalently bonded elements.) you use the name that you have memorized (however, since we only talk about a couple of polyatomic cations this should not prove to be too difficult. 2 3 4 5 6 Working out the charges of ions The charge of many ions can be worked out using patterns in the periodic table. If the cation is a "simple" cation ("simple" ion is made from only one element.) the cations is named after the element. A few of the elements in groups 3 though 13 form only one cation therefore, a few of them are type I cations-examples include Ag +, Cd 2+, Zn 2+, and Al 3+. All the elements in the first two families form type I cations. Naming compounds with type I cations-type I cations are cations that have one possible charge. Be sure to memorize silver (Ag + ) and zinc ( Zn 2 + ) charges in addition to this chart.I. 
Within a group (family) of elements, atoms form ions of a certain charge. The charge that an atom acquires when it becomes an ion is related to the structure of the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
